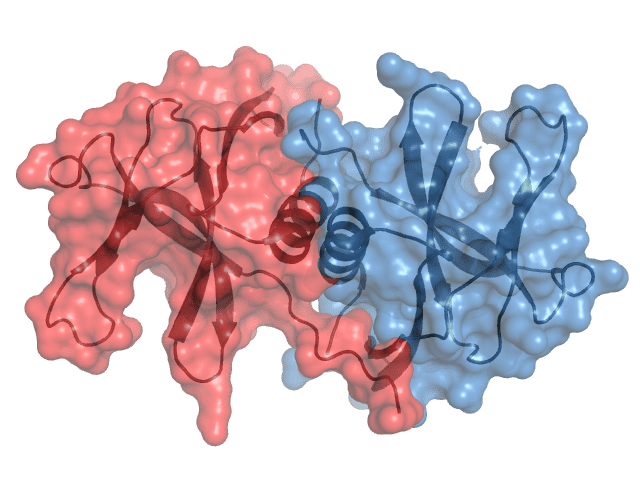
Nsp9 (ssRNA binding protein) is believed to mediate viral replication and virulence.
It has been shown that nsp9 can bind long strands of oligonucleotides (ssRNA), no direct interaction site has been identified in the protein.
Nsp9 has a unique fold not observed outside of coronaviruses; however, homologs have been found across the subfamily. It forms an obligate homodimer in solution and has been observed to form this dimer through a GxxxG alpha helical motif.
In addition, peptides featuring the cleavage motif (LEVL) of main protease (nsp5) have been shown to have binding affinity to nsp9. These peptides interact with the dimerization site structure, but do not hinder dimerization.
All in all, a mysterious protein.