Introduction
The coronavirus pandemic hit the entire world and caused millions of deaths. More than fifty companies race towards developing a vaccine to stop the disease (1). Vaccination presents a lasting solution to this unfavourable situation, reducing the burden of Coronavirus (2). The first vaccine to be approved for emergency use is an mRNA-based vaccine (3,4). How does it work? Here, I will shed some light on this, but before we go into details, we need to discuss a little about vaccines and how a foreign substance provokes an immune response in the body (immunogenicity).
Early impact of vaccines on humanity
Vaccines have existed since 1796 and have saved millions of lives over the years (5,6). Vaccines need to be given orally, intramuscularly or subcutaneously to, stimulate the body for an immune response and generate lasting immunity.
How the body fights diseases
The human body works very hard to remove any foreign substance (potential pathogens). When pathogens, such as bacteria or viruses enter the body, they attack and spread, causing disease. The immune system fights these infections recognizing a part of the pathogen using white blood cells which consist primarily of macrophages, B-lymphocytes and T-lymphocytes (7). Macrophages are antigen-presenting cells that engulf pathogens and digest them. Macrophages present parts of the pathogens to the T-lymphocytes and B-lymphocytes. When the B-lymphocytes are activated, they produce antibodies that target specific pathogens and destroy them. Also, when the T-lymphocytes are activated by the antigen-presenting cells, they seek out and destroy any infected cell in the body. However, it takes a couple of days for the body to generate the antibodies to fight an infection. After recovering from an infection, the immune system can remember how it fought the infection and can quickly in the future, prevent reinfection by the same or similar diseases. When the infection is gone, the body is left with the supply of memory cells called T-lymphocytes and B-lymphocytes that are responsible for this rapid intervention (7). Vaccines cause the same process but without a serious illness and often even without an infection. Individuals react to vaccines differently, some persons experience symptoms like swelling or redness at the injection site, low-grade fever, tiredness etc. These minor symptoms result as the body tries to build immunity against the disease, and it is your body`s healthy response. It would be no good and impracticable for the vaccine to cause the full-blown disease - instead, vaccines prime the immune system to jump into action should you ever encounter the real thing.
Traditional vaccines
Traditional vaccines contain either an attenuated (live or weakened) virus, an inactivated virus, or even a piece of a viral protein that can produce the immune answer – a so-called antigen that does not cause an actual disease but still stimulates the body to produce antibodies to fight a real infection (8). It tricks the immune system into thinking that an infection has occurred and the immune system responds by producing antibodies against the virus (9). mRNA-vaccines, however, do not contain dead or live pieces of the virus – they contain the genetic instructions necessary for our cells to make copies of the antigen itself. So, what is mRNA?
What is mRNA?
mRNA is how our body encodes information from the genome that is to be used as blueprints to make proteins: In every human cell, we find a nucleus that contains our genome in the form of DNA. This DNA is transcribed into mRNA, which then can leave the nucleus and enter the rest of the cell. DNA itself cannot leave the nucleus and this protects the cell’s genome from damage and manipulation. Once outside the nucleus, the mRNA is translated into proteins which then fulfil all kind of work tasks in our cells – they make up our hair, break down our food, transport oxygen around our body (10,11). They pretty much do everything that makes us go.
An mRNA vaccine exploits this process: When the mRNA vaccine enters our cells, it effectively skips the transcription step (DNA to RNA) and goes straight to translation (mRNA to proteins) to produce antigenic proteins (10,12).
How do the SARS-CoV-2 mRNA-based vaccines work?
The best antigen in the coronavirus SARS-CoV-2 – the bit which is recognized by the immune system – is the spike protein. The outside of the SARS-CoV-2 (Coronavirus) is carries these spike proteins, which the virus uses to enter human cells and cause an infection.
New mRNA vaccines contain messenger RNA which encodes the spike protein and as it enters the cell, the spike proteins are produced within the cell. The spike protein then migrate to the surface of the cell, where the immune system recognizes it as foreign and the body will produce antibodies to fight the infection. At this point, the process to produce immunity is the same as for any other vaccine. When infected again, the body can rapidly supply memory cells- T-lymphocytes and B-lymphocytes that remember how to attack a similar pathogen. So, when the real coronavirus SARS-CoV-2 (spike proteins and all) enters the body, the immune system remembers how it fought the formerly produced spike proteins and can easily fight off the infection.
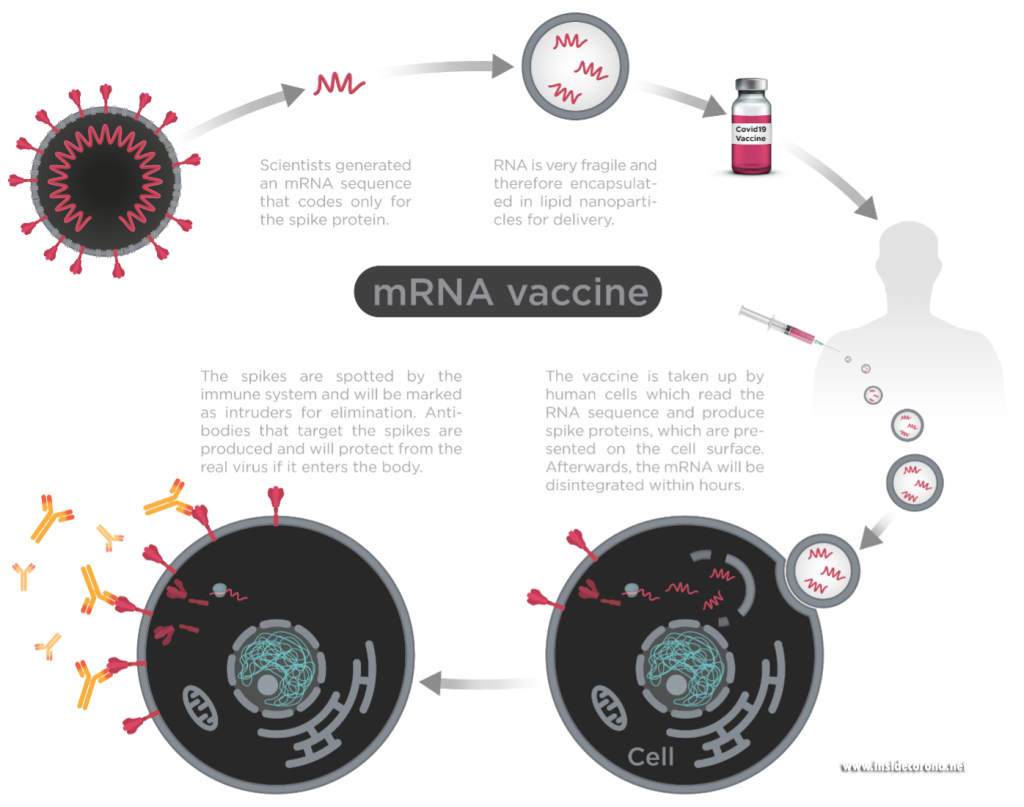
Figure1: Process of mRNA vaccines in the body.
Image by Thomas Splettstösser.
What is in the vaccine and why do we use it?
mRNA-based SARS-CoV-2 vaccines contain the mRNA strands encapsulated in lipid nanoparticles (think soap bubble) to protect it from hot temperatures as well as degrading enzymes (13,14). After being injected intramuscularly, the protection offered by the lipid nanoparticles helps the mRNA to remain active/potent until it enters the cells and releases mRNA.
There are some advantages to mRNA vaccines, and I think it can be expected that we will soon see more of them for other illnesses. The production of mRNA vaccine doses is much faster and cheaper than traditional vaccines since it does not require a long process of growing viral proteins in a cell or an egg which then needs to be deactivated or killed to produce the vaccine (15). Also, in mRNA vaccines there is never a complete virus, so contracting the disease is impossible, which can happen with live vaccines. Finally, should mutations occur, it is relatively straightforward to change the mRNA vaccine to contain this new mutation.
However, the storage temperature of an mRNA vaccine is a challenge: they need to be stored at a very low temperature to maintain its potency until it is ready to be used and this type of freezer is not available everywhere and makes transport difficult compared to a traditional vaccine (14,16).

Figure2: Sample storage freezer for mRNA vaccines.
Image by Yunyun Gao.
Can an mRNA-based vaccine change my DNA?
As the vaccine contains no so-called “reverse-transcriptase”, the spike mRNA cannot be converted into DNA. mRNA-based vaccines cannot even enter the cell’s nucleus. Hence, they are not able to change your DNA. They are just a messenger to produce the spike protein and the enzymes of the cell readily destroy them shortly afterwards. Also, the unfortunate cell itself is destroyed by the immune system once it displays the spike on the surface and the immune system has learned to recognize it.
Do I still need to wear masks?
Also, remember that after getting vaccinated, your body needs time to develop antibodies in sufficient quantities, so you can still contract Coronavirus even after being vaccinated. Hence, we need to maintain safety measures (social distancing, masking, handwashing etc) while allowing immunity to develop after being vaccinated. It is also still unclear what happens if an infected person is vaccinated, or in people who have no strong immune system (immunocompromised patients). “I have been vaccinated, can I now go, hugging and kissing on the street?” Sorry, you can’t. This is because you can still contract the virus even after being vaccinated: the body needs time to develop immunity against an infection. This is the main reason for jab-spacing as it enables the body to build an immune response.
Conclusion
Vaccines trigger the production of memory cells (T-lymphocytes and B-lymphocytes) to fight infections and can protect us from life-threatening diseases. The more people get vaccinated, the more likely we are to achieve herd-immunity, where even unvaccinated people are protected. A good example of herd-immunity is a burning bush. The fire keeps spreading but when it encounters a large space or a river, it stops spreading and the other side of the bush will not be affected. However, the space must be large enough to make this happen. Vaccinated individuals are like the large space or river, they help in stopping the spread of the disease to the unvaccinated hence, protecting the unvaccinated. Also, remember that for this kind of protection to occur, a sufficient number of individuals must have been vaccinated. There is no scientific basis to show that an mRNA-based vaccine can change your DNA. Developing an mRNA vaccine within a short period is a phenomenal advancement in science. As people are being vaccinated, every other protective measure is encouraged if we want to end this pandemic. We can only think of relaxing measures when the number of cases has considerably reduced. Stay safe and healthy, and together we are protected.
Acknowledgement
I would like to thank Dr Andrea Thorn, Dr Dale Tronrud, Dr Sam Horrell and Dr Yunyun Gao for their wonderful and helpful suggestions. Also, my thanks go to Dr Thomas Splettstoesser for making an image used for this post.
References
1. Regulatory Affairs Professionals Society. COVID-19 vaccine tracker [Internet]. [cited 2021 Jan 10]. Available from: https://www.raps.org/news-and-articles/news-articles/2020/3/covid-19-vaccine-tracker
2. Nature Communications. Vaccines work. Nat Commun [Internet]. 2018 Apr 24 [cited 2021 Jan 3];9. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5915378/
3. FDA. COVID-19 Vaccines. FDA [Internet]. 2021 Feb 18 [cited 2021 Feb 19]; Available from: https://www.fda.gov/emergency-preparedness-and-response/coronavirus-disease-2019-covid-19/covid-19-vaccines
4. Mueller B. U.K. Approves Pfizer Coronavirus Vaccine, a First in the West. The New York Times [Internet]. 2020 Dec 2 [cited 2021 Feb 19]; Available from: https://www.nytimes.com/2020/12/02/world/europe/pfizer-coronavirus-vaccine-approved-uk.html
5. World Health Organization. Vaccines and immunization [Internet]. [cited 2021 Jan 10]. Available from: https://www.who.int/health-topics/vaccines-and-immunization#tab=tab_1
6. Thèves C, Biagini P, Crubézy E. The rediscovery of smallpox. Clinical Microbiology and Infection. 2014 Mar;20(3):210–8.
7. Vitetta ES, Berton MT, Burger C, Kepron M, Lee WT, Yin XM. Memory B and T Cells. Annual Review of Immunology. 1991;9(1):193–217.
8. Centers for Disease Control and Prevention. Basics of Vaccines | CDC [Internet]. 2019 [cited 2021 Jan 10]. Available from: https://www.cdc.gov/vaccines/vpd/vpd-vac-basics.html
9. Sell S. How vaccines work: immune effector mechanisms and designer vaccines. Expert Rev Vaccines. 2019 Oct;18(10):993–1015.
10. Pardi N, Hogan MJ, Porter FW, Weissman D. mRNA vaccines — a new era in vaccinology. Nature Reviews Drug Discovery. 2018 Apr 1;17(4):261–79.
11. Zhang C, Maruggi G, Shan H, Li J. Advances in mRNA Vaccines for Infectious Diseases. Front Immunol [Internet]. 2019 Mar 27 [cited 2021 Jan 11];10. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6446947/
12. Jackson NAC, Kester KE, Casimiro D, Gurunathan S, DeRosa F. The promise of mRNA vaccines: a biotech and industrial perspective. npj Vaccines. 2020 Feb 4;5(1):11.
13. Martin C, Lowery D. mRNA vaccines: intellectual property landscape. Nat Rev Drug Discov. 2020 Sep;19(9):578–578.
14. Baden LR, Sahly HME, Essink B, Kotloff K, Frey S, Novak R, et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. New England Journal of Medicine [Internet]. 2020 Dec 30 [cited 2021 Jan 10]; Available from: https://www.nejm.org/doi/10.1056/NEJMoa2035389
15. Sandbrink JB, Shattock RJ. RNA Vaccines: A Suitable Platform for Tackling Emerging Pandemics? Front Immunol. 2020;11:608460.
16. Pan American Health Organization WHO. COVID-19 Vaccine Explainer: COMIRNATY®, COVID-19 mRNA vaccine - PAHO/WHO | Pan American Health Organization [Internet]. [cited 2021 Feb 5]. Available from: https://www.paho.org/en/documents/covid-19-vaccine-explainer-comirnatyr-covid-19-mrna-vaccine
Overview
During the Corona-dominated year 2020 scientists all over the world united and gathered as much information as possible to understand the exact mechanism behind the lifecycle of SARS-CoV-2.
The main question was: how can we stop the virus from invading the human cell and causing COVID-19? A focus in the quest to answer this question, was the SARS-CoV-2 entry mechanism. The group of Janet Iwasa contributes to this ongoing research process by providing a high-quality video animation of the SARS-CoV-2 entry into the human host cell. This current version of the entry animation has already been shown on PBS News (08.12.20) and we aim to improve it with your help in 2021 (see below)!
The Entry Animation
Click this Link to see the Entry Animation on YouTube.
This entry animation is a collection of current knowledge about the SARS-CoV-2 entry mechanism. What we know at this point is that the mechanism starts with the viral approach. An individual can be infected with SARS-CoV-2 after inhaling airborne viral particles. These viruses can then travel into the airways, where they may encounter host cells of the respiratory epithelium in the trachea and lungs.
As you can read in a previous blogpost, the Spikes (teal) are Corona’s key to invade the host cell and thus of great interest in terms of vaccination and therapeutic approaches against COVID-19. The Spike protein recognizes a specific receptor on the human host cell surface, called ACE2 (purple). Usually, the Spikes are very dynamic and able to undergo opening, closing and bending movements. But after binding to ACE2, the protein is locked into its open position. Another protein on the cell surface, called TMPRSS2 (orange), can then come along and cut the Spike protein in a specific location. These segments of the Spike protein fall away, exposing a portion of the Spike protein which was previously hidden.
The Spike protein is then able to undergo a series of dramatic conformational changes. During the first stage, the Spike protein inserts itself into the membrane of the cell. In the second stage, segments of the Spike protein zipper back on itself, forcing the membrane of the cell and the viral membrane to fuse. After fusion, the viral RNA is deposited into the host cell, where it will direct the cell to produce more virions. This process is known as post-fusion.
The Annotation Tool
Click this Link to use the Annotation Tool.
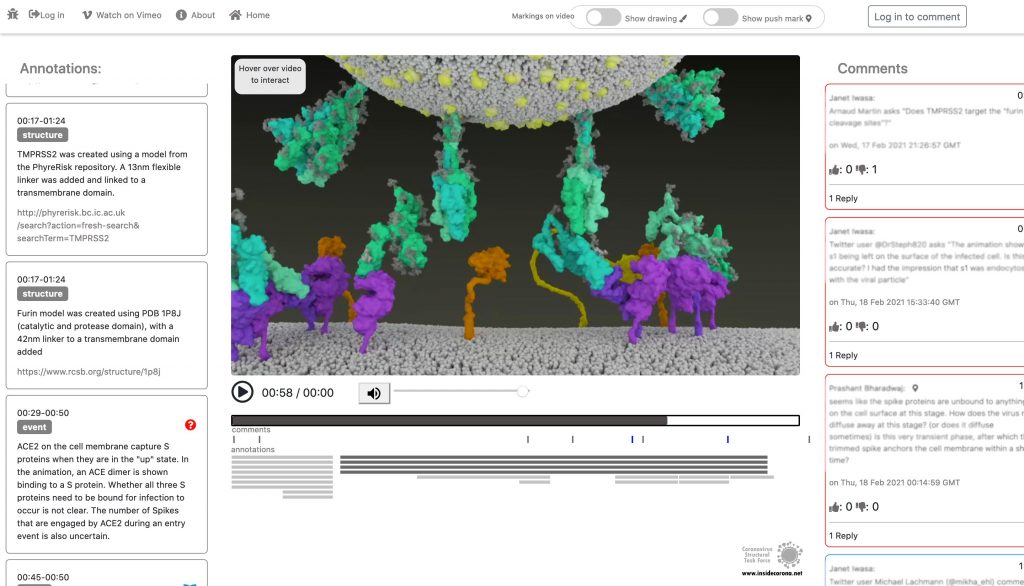
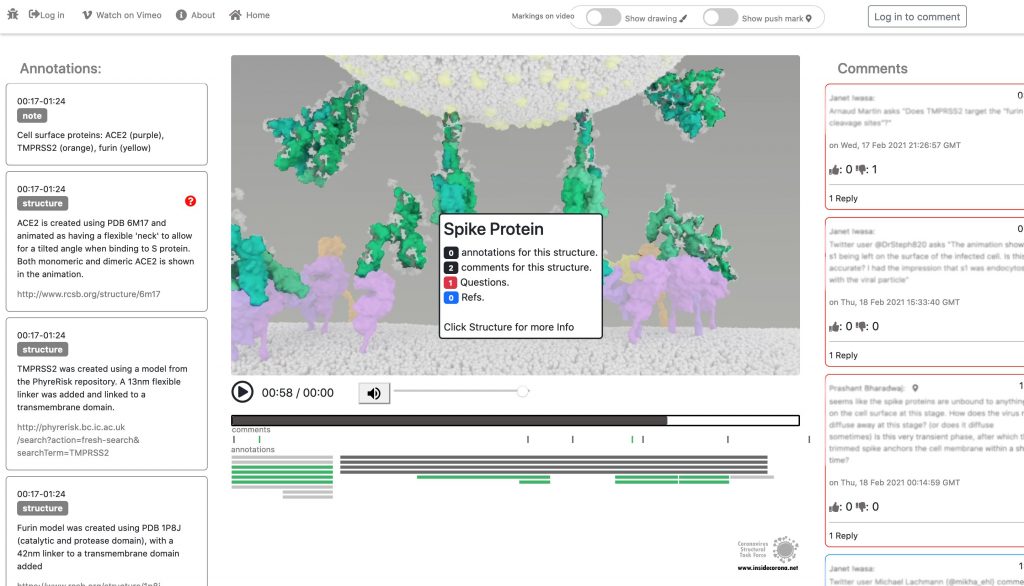
In January, this will be supplemented with a tool so that the knowledge about the SARS-CoV-2 entry mechanism can be discussed interactively by scientists all over the world. This online platform will serve as a basis for scientific discussion by providing an annotation tool. Scientific users can set a pin at any point of the video and comment their suggestions, criticism or questions about the mechanism and the structure depictions (see Fig. 1 for a prototype). Based on these annotations, the Iwasa Group will improve the animation of the entry process to provide an up-to-date detailed representation of this key process. The resulting entry animation is not only addressed to scientists, but it is also used for public outreach and education.
Even though the entry mechanism is not entirely understood yet, it could already be depicted in the fantastic animation of the Iwasa Group. There are still a lot of details and additional information to be found out about this process. From January on, the annotation tool therefore will provide the opportunity to discuss this mechanism publicly.
Thanks to the Iwasa Group for this Christmas present!
Merry Christmas!
Introduction
It is known as VUI‑202012/01 or B.1.1.7 – the new mutation of the coronavirus Sars-CoV-2. It may be responsible for a sharply increased number of infections in the southeast of England (1), however, the scientific results leading to very strict lockdown measurements in the south of the UK, and travel restrictions across Europe are few and far between. Here, we have compiled what is known up until now.
On mutations
Mutations are normal in the evolution of life – and of viruses. If two similar viruses have infected the same cell, their genomes can become mixed-up, one of the reasons why animal influenza strains are considered so dangerous. This is also called recombination. Mutations can be caused by chemicals, radiation (including UV light) and errors during genome copying. A typical SARS-CoV-2 virus accumulates two amino acid changes per month in its genome — a rate of change about half that of influenza (2). This is because SARS-CoV-2 can repair RNA to some extent. But even so, this natural process led to thousands of mutations since the beginning of the pandemic. If they affected the virus life cycle negatively, that strain may have likely died out - if they did not make a difference or enhanced its chances of survival, it may have persisted.
Many mutations that are observed occur in the spike protein, which both serves to recognize potential host cells but is also what is being recognized by antibodies (i.e., the immune system).
Changes here can be crucial for the survival of the virus (“evolutionary pressure”) as they could significantly alter its affinity to the human receptor ACE2, which the virus uses as gateway to our cells.
What vaccines do
Most, if not all, potential COVID-19 vaccines expose our body to some part of the spike protein, which can be made by the body itself (mRNA vaccines) or carried by a harmless virus instead of SARS-CoV-2 (vector). Our body then produces antibodies which specifically recognize the spike and persist for several months. If we are exposed afterwards to the real virus, the body can recognize it immediately – and the risk of infection is much lower as the immune system swings into action immediately. Earlier this year, the spike mutation D614G (amino acid residue number 614 changing from aspartic acid (D) to glycine (G)) caused quite a stir in the media, and became the predominant form of SARS-CoV-2 (2, 3). However, if and in how far this was caused by natural selection is still debated (3). Another example which triggered an increased media coverage was the mutation Spike Y453F, which originated from infected minks in Denmark (4) and led to a culling of millions of animals. In any case, if we would be vaccinated with a spike protein form that would be different from the one in a virus we encounter later, there is a small chance that the vaccine may be rendered ineffective. This chance is, however, small for SARS-CoV-2, in any case much smaller than for HIV, which famously evaded any attempt to develop a vaccine.

What do we know?
There was a steep rise in infections in the UK recently, as in most other European countries.
A new mutation of the virus has emerged and seems to replace the old version of SARS-CoV-2 (5). Thousands of patients have been found to carry this variant.
This new variant has more mutations at once than expected. These mutations have not observed in this combination before.
The variant has been reported in the UK, the Netherlands, Denmark, Australia and Belgium so far.
What is striking to me as scientist about these findings is one thing in particular: How could the British government find that thousands of people were having the new SARS-CoV-2 variant, instead of the old, if the illness does not look any different? Sequencing samples from each and every patient would be technically very challenging, if not impossible. How could they know? The answer is:
Serendipity
The main PCR test employed in the United Kingdom is Thermo Fisher's TaqPathCOVID-19. This test identifies RNA on three different genome locations: In ORF1ab, nucleotide and spike. Now, it stopped working for the spike portion of the test, while the other two RNAs were still found to be present, which likely prompted scientists to sequence some of the samples in question. And indeed, the new mutant has a deletion of histidine-69 and valine-70, called 69-70del. This permitted easy differentiation of patients with the old SARS-CoV-2 (3 hits) and the new (2 hits) and is the reason why we know so much about the epidemiology of this variant!* It has also to be said that this test is not used as often in other countries, such as Germany, and this could well be the reason why we do not know if and how widespread it is here. In addition, other countries sequence much smaller proportions of virus isolates than the UK, so ongoing circulation of this variant outside of the UK cannot be excluded.
The details of the mutation
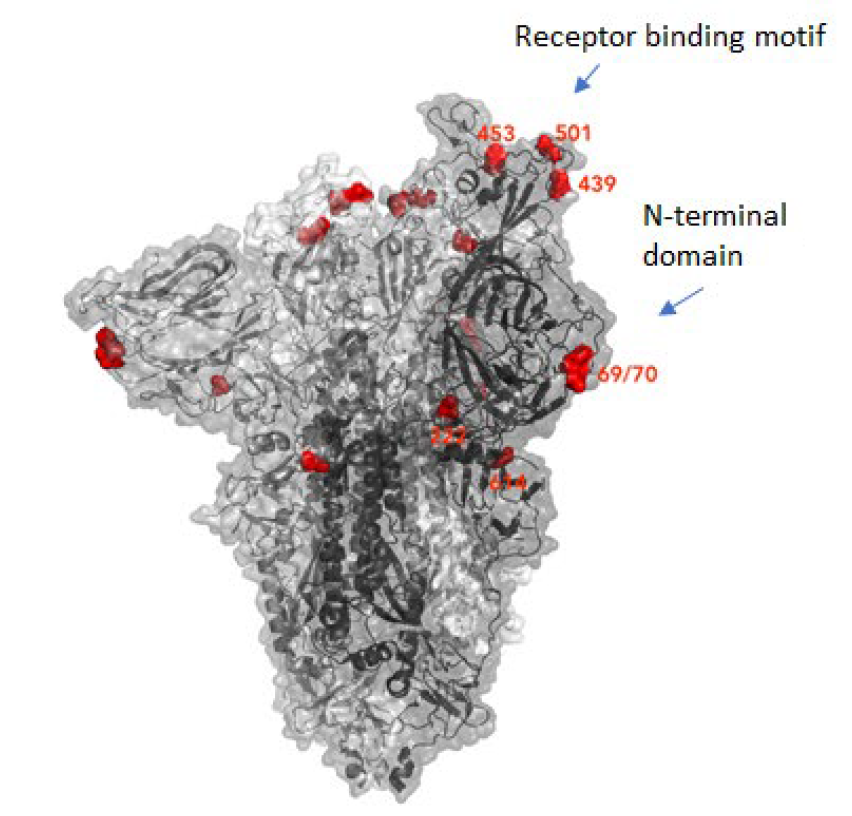
The new variant of SARS-CoV-2 VUI-202012/01 has 14 amino acid changes and three deletions affecting the genes for ORF1ab, spike and ORF8. One of these mutations (N501Y) occurs in the receptor binding domain and could lead to an increased binding affinity to the human ACE2. The 69-70 deletion has likely an immunological role and is the reason this mutant was detected so widely, as this RNA location is used for PCR tests. Another interesting mutation is the P681H, which is next to a furin cleavage site that has a biological significance in membrane fusion. These mutations could be responsible for the increased transmissibility. The effects of the other mutations aren’t fully investigated yet. Here is a list of the mutations which have been observed in the VUI‑202012/01 or B.1.1.7 variant:
| T1001I in gene ORF1ab | |
| A1708D in gene ORF1ab | |
| I2230T in gene ORF1ab | |
| SGF 3675-3677 deletion in gene ORF1ab | |
| A1708D in gene ORF1ab | |
| HV 69-70 deletion in spike | The 69-70 deletion on the spike protein is a re-occurring mutation that has shown to often co-occur with other amino acid changes in the RBD (6, 7). (1) Evasion to the human immune response and in association with other receptor binding domain changes (1) (2) Immunological role (8) (3) Leads to diagnostic failures which permit detection (see above, "Serendipity") (4) Associated with immune escape in immunocompromised patients (9(8)) Furthermore, the 69-70 deletion arose in multiple unrelated lineages and is associated with the evasion of the immune response (9). It is being hypothesized that this mutation undergoes a strong positive selection when exposed to convalescent plasma therapy in an immunocompromised human host (7). |
| Y144 deletion in spike | Deletion in the spike N-terminal domain (9) |
| N501Y in spike | One of six key contact residues in the spike receptor binding domains, this mutation leads to an increasing binding affinity to human and murine ACE2 (1). |
| A570D in spike | Mutation located at the spike receptor binding domain (10) |
| P681H in spike | The P681H mutation is located directly next to the furin cleavage site. It is one of the four residues which are insertions when compared to closely related coronaviruses, creating a furin cleavage site in the spike protein between the spike S1 and S2 domains. This prompts the entry of the virus into respiratory epithelial cells as well as the transmission in animal models (1) The S1/S2 furin cleavage site of SARS-CoV-2 is not found in closely related coronaviruses and has been shown to promote entry into respiratory epithelial cells and transmission in animal models (9) |
| T716I in spike | Mutation in in the S2 domain |
| S982A in spike | Mutation in in the S2 domain (10) |
| D1118H in spike | Mutation in in the S2 domain (8) |
| Q27 stop in ORF8 | The Q27stop mutation in the ORF8 leads to the truncation of the ORF8, and as it only consists of 121 amino acids, the consequence might be a loss of function. These and the other mutations could be responsible for the increased transmissibility of the B.1.1.7 variant. In any case, this mutation truncates the ORF8 protein at residue 27 or renders it inactive which allows further downstream mutations to accrue. (1) |
| R52I in ORF8 | |
| Y73C in ORF8 | |
| D3L in nucleocapsid | |
| S235F in nucleocapsid |
Why were there so many mutations at once?
This could be a result of prolonged or chronical SARS-CoV-2 infections as study of these infections reveal unusually large numbers of nucleotide changes and deletion mutations and often high ratios of non-synonymous changes. In addition to this, convalescent plasma treatment can cause intra-patient virus genetic diversity (11).
What does the new mutation mean in terms of impact and epidemiology?
There was an increase in cases with the new strain in total and in
proportion to the old (1). What does that mean for us?
This is what the internet says:
The COVID-19 genomics UK consortium (COG) reports about a “priority set of SARS-CoV-2 Spike mutations that are of particular interest based on potential epidemiological significance in the UK and/or biological evidence based on the literature or unpublished work.” (9)
The New and Emerging Respiratory Virus Threats Advisory Group of the British government (NERVTAG) discussed the new variant on Friday and concluded that its growth rate is higher by 67-75% and that this is likely due to a selective advantage. “In summary, NERVTAG has moderate confidence that VUI-202012/01 demonstrates a substantial increase in transmissibility compared to other variants.” (12) This is very likely the source of Boris Johnson’s claim to this strain being “70% more infectious”.
The English government writes that PHE (Public Health England) „is working with partners to investigate and plans to share its findings over the next 2 weeks. There is currently no evidence to suggest that the variant has any impact on disease severity, antibody response or vaccine efficacy. High numbers of cases of the variant virus have been observed in some areas where there is also a high incidence of COVID-19. It is not yet known whether the variant is responsible for these increased numbers of cases.” (13)
Conclusion
From this, we conclude that the British government, and we, do not know yet. It has not been conclusively shown that the new variant is more infectious (likely), has an easier time to evade the host immune system or if the vaccine will be less effective against it (very unlikely). The epidemologic model which predicts a higher tranmissability has still to be published, the science is still in the making. Tests of vaccines against the new variant are ongoing and will take a few weeks. There is yet little evidence that this new variant poses a significantly bigger threat than others - or to the contrary.
Acknowledgements
While I am listed as author of this article, it could not have been written without the help and research by Pairoh Seeliger, Lea von Soosten, Luise Kandler, Erik Nebelung and Oliver Kippes who all helped in this.
I would also thank Nicolai Wilk from Thermo Fisher Scientific who quickly responded to my questions about their test.
The title picture shows mutation cards from the game Pandemic Expansion: On the Brink by Z-Man Games.
- *The 69-70del mutation is predominantly observed in B.1.1 (including B.1.1.7), B.1.258, and the cluster 5 variant lineages of SARS-CoV-2.
References
- 1.A. Rambaut, Preliminary genomic characterisation of an emergent SARS-CoV-2 lineage in the UK defined by a novel set of spike mutations. virological.org (2020), (available at https://virological.org/t/preliminary-genomic-characterisation-of-an-emergent-sars-cov-2-lineage-in-the-uk-defined-by-a-novel-set-of-spike-mutations/563).
- 2.E. Callaway, The coronavirus is mutating — does it matter? Nature, 174–177 (2020).
- 3.L. Zhang, C. B. Jackson, H. Mou, A. Ojha, H. Peng, B. D. Quinlan, E. S. Rangarajan, A. Pan, A. Vanderheiden, M. S. Suthar, W. Li, T. Izard, C. Rader, M. Farzan, H. Choe, SARS-CoV-2 spike-protein D614G mutation increases virion spike density and infectivity. Nat Commun (2020), doi:10.1038/s41467-020-19808-4.
- 4.ECDC, Detection of new SARS-CoV-2 variants related to mink. www.ecdc.europa.eu (2020), (available at https://www.ecdc.europa.eu/sites/default/files/documents/RRA-SARS-CoV-2-in-mink-12-nov-2020.pdf).
- 5.ONS UK , Percentage of COVID-19 cases that are positive for ORF1ab and N genes. www.ons.gov.uk (2020), (available at https://www.ons.gov.uk/peoplepopulationandcommunity/healthandsocialcare/conditionsanddiseases/adhocs/12690percentageofcovid19casesthatarepositivefororf1abandngenes).
- 6.R. M. Dawood, M. A. El-Meguid, G. M. Salum, K. El-Wakeel, M. Shemis, M. K. El Awady, Bioinformatics prediction of B and T cell epitopes within the spike and nucleocapsid proteins of SARS-CoV2. Journal of Infection and Public Health (2020), doi:10.1016/j.jiph.2020.12.006.
- 7.S. A. Kemp, D. A. Collier, R. Datir, S. Gayed, A. Jahun, M. Hosmillo, I. A. Ferreira, C. Rees-Spear, P. Mlcochova, I. U. Lumb, D. Roberts, A. Chandra, N. Temperton, K. Sharrocks, E. Blane, J. A. Briggs, K. G. Smith, J. R. Bradley, C. Smith, R. Goldstein, I. G. Goodfellow, A. Smielewska, J. P. Skittrall, T. Gouliouris, E. Gkrania-Klotsas, C. J. Illingworth, L. E. McCoy, R. K. Gupta, Neutralising antibodies drive Spike mediated SARS-CoV-2 evasion (2020), , doi:10.1101/2020.12.05.20241927.
- 8.K. Kupferschmidt, Mutant coronavirus in the United Kingdom sets off alarms, but its importance remains unclear. Science (2020), doi:10.1126/science.abg2626.
- 9.COG, COG-UK update on SARS-CoV-2 Spike mutations of special interest Report 1. https://www.cogconsortium.uk (2020), (available at https://www.cogconsortium.uk/wp-content/uploads/2020/12/Report-1_COG-UK_19-December-2020_SARS-CoV-2-Mutations.pdf).
- 10.S. Kemp, W. Harvey, R. Datir, D. Collier, I. Ferreira, A. Carabelii, D. L. Robertson, R. K. Gupta, Recurrent emergence and transmission of a SARS-CoV-2 Spike deletion ΔH69/V70 (2020), , doi:10.1101/2020.12.14.422555.
- 11.ECDC, Threat Assessment Brief: Rapid increase of a SARS-CoV-2 variant with multiple spike protein mutations observed in the United Kingdom. www.ecdc.europa.eu (2020), (available at https://www.ecdc.europa.eu/en/publications-data/threat-assessment-brief-rapid-increase-sars-cov-2-variant-united-kingdom).
- 12.NERVTAG, NERVTAG meeting on SARS-CoV-2 variant under investigation VUI-202012/01. https://khub.net (2020), (available at https://khub.net/documents/135939561/338928724/SARS-CoV-2+variant+under+investigation%2C+meeting+minutes.pdf/962e866b-161f-2fd5-1030-32b6ab467896?t=1608470511452).
- 13.PHE, PHE investigating a novel variant of COVID-19 . www.gov.uk (2020), (available at https://www.gov.uk/government/news/phe-investigating-a-novel-variant-of-covid-19).
COVID-19 is caused by the new coronavirus SARS-CoV-2. This virus has a characteristic virus hull featuring surface proteins which are commonly called “spikes”. Protruding from the viral hull like “spikes of a crown”, they give the coronavirus its name (corona = crown). These proteins make the first contact with human cells and are akin to keys that use a human receptor called “angiotensin-converting enzyme2” (ACE2) as a backdoor to gain access to and infect the cell.
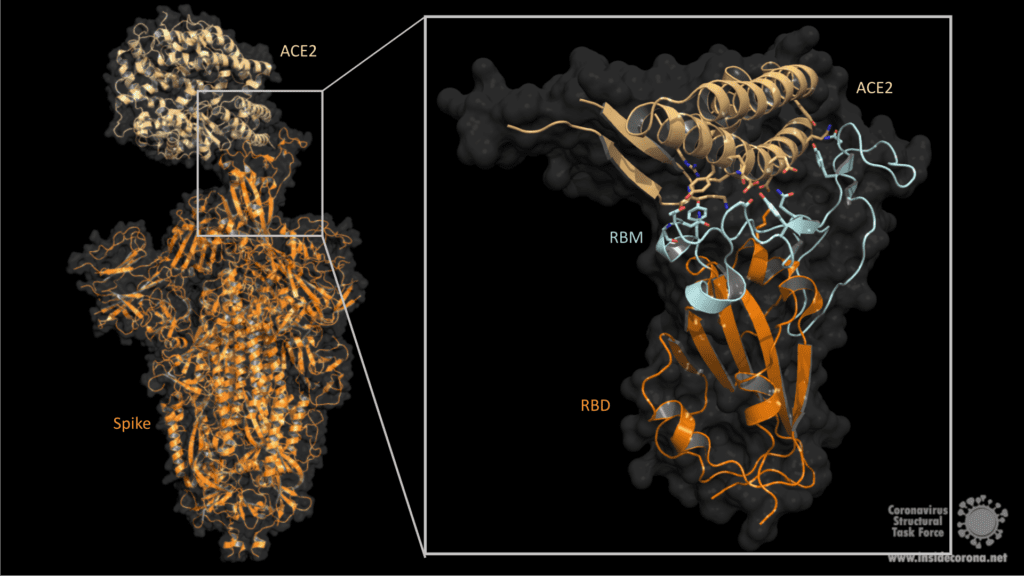
1. Fuction of ACE2
ACE2 is a membrane protein which is anchored in the human cell membrane of epithelial cells. This type of cells can be found on the surface of lung, intestine, heart and kidney tissue. As a type I membrane protein, its primary function is to take part in maturation of angiotensin, a peptide hormone which controls vasoconstriction and blood pressure. ACE2 can be compared to a lock which can be unlocked by the coronavirus spike protein. The virus can then enter the cell and hijack its functions to reproduce itself, thus causing the Covid-19 infection which poses a serious danger to humanity, especially for older people and people with pre-existing conditions. For this reason, one approach to combating SARS-CoV-2 is to target and inhibit the spike to prevent infection. In order to do so, knowledge of the structural features of the spike and its interaction processes with ACE2 are indispensable. (Further information about how macromolecular structures are visualized can be found on our homepage: https://insidecorona.blogs.uni-hamburg.de/visualizing-macromolecular-structures/)
2. Spike: Structure and Fusion Mechanism
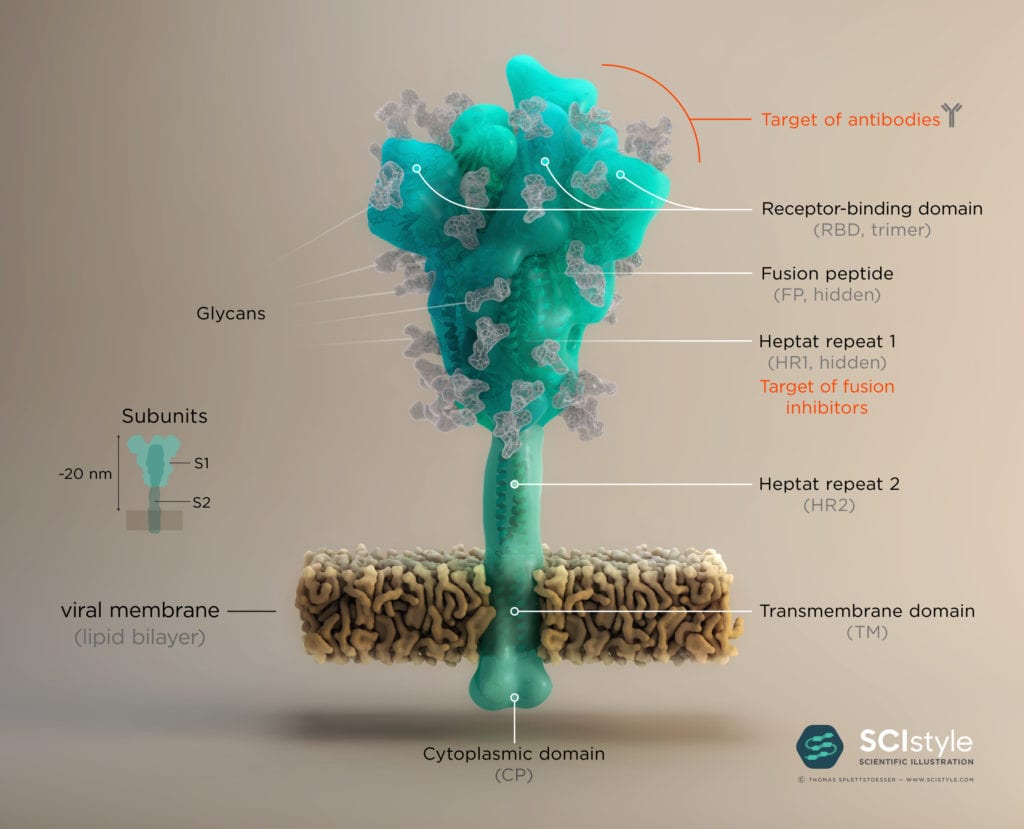
The Spike protein has a trimeric shape comprising three identical monomeric structural elements. Each of these monomers can fold out akin to a modern car key with a fold-out key element with specific teeth on its surface. This fold-out key element is the so-called “receptor binding domain” (RBD). The spike can only interact with ACE2 when its RBD is in a folded-out position, exposing its teeth, or “receptor binding motive” (RBM). As the name suggests, it comprises a motive of different amino acids which then can bind and unlock the ACE2 receptor. This key lock mechanism triggers a cascade of events initiating fusion with the host cell. First, protein scissors are recruited to the binding site. These scissors (furin & transmembrane serine protease 2) cleave the spike protein for subsequent activation. The active spike molecule then rearranges itself to form a long structural “hook” (formed of HR1/ HR2 and FP see Fig.2) that brings the epithelial cell and viral cell membrane into close proximity for fusion. Once the fusion is completed, the path for the virus is clear to transfer its genome encoded in ribonucleic acid (RNA) into the host cell. This successful transfer then enables the virus to multiply itself and finally spread from cell to cell, causeing Covid-19 in its wake.
3. Evading the Immune System with Carbohydrate Chains
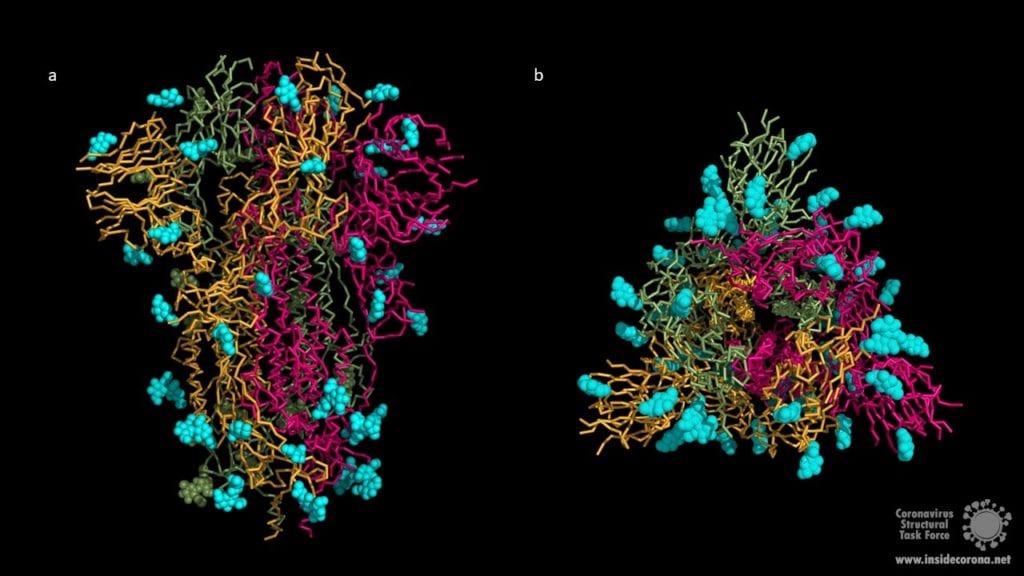
The human immune system normally recognizes the surface proteins of foreign organisms such as viruses or bacteria and reacts with an immune response to combat them. Spike proteins are such surface proteins but because of structural peculiarities, the coronavirus evades both the innate and the adaptive human immune system. The secret of these structural peculiarities are the N-glycans. These are long carbohydrate chains which sit on spike’s surface. Each spike comprises 66 N-glycans forming a protective shield around the protein. Hence the human immune system has problems recognizing spikes and identifying the coronavirus as an enemy.
The COVID 19 pandemic has a massive impact on our lives, our health and the global economy. Scientists around the world are trying to develop new drugs to combat the virus. Since the spike plays a critical role in the infection process, it is a prime target for drug development against the pandemic. One drug approach to inhibit the interaction between spike and the ACE2 receptor is to cap the spike protein using antibodies. Antibodies are proteins, normally produced by the human immune system to fight viruses. The idea is to treat patients with antibodies that cap the RBD of spike, thus preventing interactions with ACE2. This would lead to a nonfunctional spike, blocking the coronavirus from entering the cell (The key would no longer fit the lock). Another approach includes the development of small molecules that target and inactivate the protein scissor transmembrane serine protease 2 (see chapter 2), as the spike’s functionality depends on its cleavage activity. Since the spike protein decorates the virus hull, it could even be part of a potential vaccine. For this reason, the spike protein could also become the key in the molecular fight against COVID-19.
