EnoRNase (also known as NendoU, nsp15 or nidoviral RNA uridylate‐specific endoribonuclease) is a viral enzyme that cuts at uridine in the middle of an RNA sequence, which results in a 2′‐3′ cyclic phosphodiester and 5′‐hydroxyl terminus.
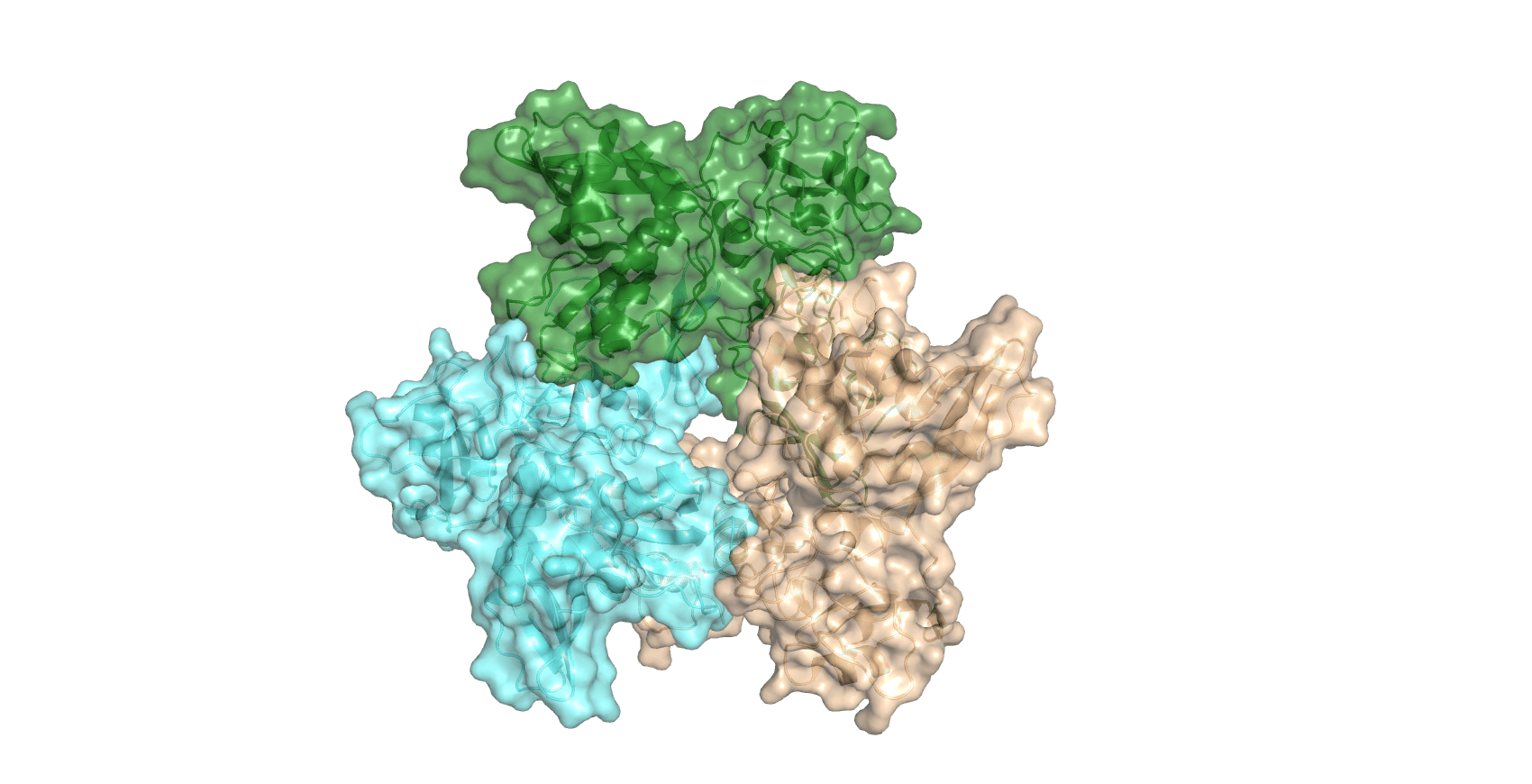
One nsp15 monomer is made up of three distinct domains, the N-terminal oligomerisation domain, a middle domain, and the catalytic NendoU domain at the C-terminus.
Six monomers then form a double-ring hexamer stabilised by an N-terminal oligomerisation domain, which is the functional form.
Nsp15 is a Mn2+-dependent endoribonuclease, meaning it relies on the coordination of the metal manganese in its active site to perform the cleavage.
The exact function of this cleavage, and of nsp15 as a whole is unknown, with conflicting studies suggesting it is and isn't vital for replication.